
How stimulation intensity affects motor learning
 based on reviews by Charlotte Wiltshire and 1 anonymous reviewer
based on reviews by Charlotte Wiltshire and 1 anonymous reviewer
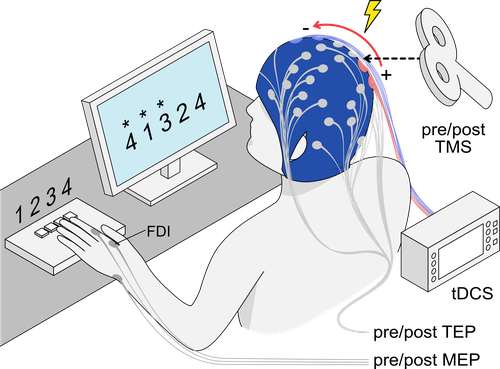
Dose-response of tDCS effects on motor learning and cortical excitability: a preregistered study
Abstract
Recommendation: posted 04 June 2024, validated 05 June 2024
Artemenko, C. (2024) How stimulation intensity affects motor learning. Peer Community in Registered Reports, . https://rr.peercommunityin.org/articles/rec?id=656
Related stage 2 preprints:
Gavin Hsu, Zhenous Hadi Jafari, Abdelrahman Ahmed, Dylan J. Edwards, Leonardo G. Cohen, Lucas C. Parra
https://osf.io/a42uy
Recommendation
Level of bias control achieved: Level 2. At least some data/evidence that will be used to answer the research question has been accessed and partially observed by the authors, but the authors certify that they have not yet observed the key variables within the data that will be used to answer the research question.
List of eligible PCI RR-friendly journals:
The recommender in charge of the evaluation of the article and the reviewers declared that they have no conflict of interest (as defined in the code of conduct of PCI) with the authors or with the content of the article.
Evaluation round #2
DOI or URL of the report: https://osf.io/cwb6p
Version of the report: 1.2
Author's Reply, 22 May 2024
Decision by Christina Artemenko , posted 15 May 2024, validated 16 May 2024
, posted 15 May 2024, validated 16 May 2024
Dear Gavin Hsu,
Thank you for resubmitting a revision of your registered report. The reviewers had a close look at the revision and were mostly satisfied. Thus, I would like to invite a revision of your registered report by taking into account the last remaining points of the reviewer and, additionally, one more formal point: Please write in the analysis section or pilot data section whether or not the pilot data will be included in final data set for analyses of the current study or not.
Thank you for submitting your registered report to PCI-RR. We are looking forward to receive your minor revisions.
Best regards, Christina Artemenko
Reviewed by anonymous reviewer 1, 18 Apr 2024
The authors have addressed my concerns.
https://doi.org/10.24072/pci.rr.100656.rev21Reviewed by Charlotte Wiltshire , 15 May 2024
, 15 May 2024
Comments have been addressed comprehensively. I still have a few comments, but I view these as minor and serve as suggestions, only. I look forward to seeing the results when they appear.
There is now added discussion of the null-effects reported in studies with similar designs. I appreciate the desire to not stoke more controversy! However, I do think it’s an important issue to tackle. There are reports of null findings at low intensities as well as intermediate intensities. I’m unaware of any at higher intensities as used here. One prediction would be that higher intensities may facilitate the likelihood of finding an effect over and above low- and intermediate- intensities (as we suggest in Wiltshire et al., 2020). This would, in my mind, provide further justification for this work.
This is just a suggestion, feel free to leave it as is. I provide our reference as it helps me to expand on the point that I'm trying to make, but it does not need to be included in the text, of course!
“Some null results of tDCS on MEP may be explained as reversal of effects at higher intensities 28 leading to a “no man’s land” at intermediate intensities[29]”
I’m still not completely convinced by the typing task. I still think that this represents touch-typing proficiency rather than any measure of general dexterity within the hands. Justification has been included to show a relationship between typing speed and motor learning outcomes, but I don’t think this justifies conceptualising the task as a measure of baseline motor dexterity. Perhaps consider rephrasing or adding this caveat – this is a suggestion only.
Rossi et al., (2020) will help to justify that combining tDCS with TMS is within safety limits (to an extent). This is an update from the 2009 paper already cited. See ref below, section 3.3 of paper.
The analysis of the right hemisphere is listed as exploratory, but I still think there seems like a prediction here – that you will use it as a within-subject control. Therefore, an analysis plan could be added and specified in advance.
I’d like to see the description of how MEPs were included/excluded included in the text (as supplementary materials, perhaps). The following text is not enough to enable replication, however the text included in the response, would be sufficient. “The average will only include trials with a detectable biphasic MEP.”
Rossi, S., Antal, A., Bestmann, S., Bikson, M., Brewer, C., Brockmöller, J., Carpenter, L. L., Cincotta, M., Chen, R., Daskalakis, J. D., Di Lazzaro, V., Fox, M. D., George, M. S., Gilbert, D., Kimiskidis, V. K., Koch, G., Ilmoniemi, R. J., Pascal Lefaucheur, J., Leocani, L., … Hallett, M. (2020). Safety and recommendations for TMS use in healthy subjects and patient populations, with updates on training, ethical and regulatory issues: Expert Guidelines. Clinical Neurophysiology. https://doi.org/10.1016/j.clinph.2020.10.003
Wiltshire, C. E. E., & Watkins, K. E. (2020). Failure of tDCS to modulate motor excitability and speech motor learning. Neuropsychologia, 107568. https://doi.org/10.1016/j.neuropsychologia.2020.107568
https://doi.org/10.24072/pci.rr.100656.rev22
Evaluation round #1
DOI or URL of the report: https://osf.io/nz2ja
Version of the report: 1.1
Author's Reply, 09 Apr 2024
Decision by Christina Artemenko , posted 07 Mar 2024, validated 07 Mar 2024
, posted 07 Mar 2024, validated 07 Mar 2024
Dear Lucas Parra,
Thank you for submitting your Registered Report on "Dose-response of tDCS effects on motor learning and cortical excitability" to PCI-RR. I received two detailed reviews that found your study of interest and raised a couple of points, such as fatigue, MEPs/TEPs, safety protocols, typing test, lateralization etc.. I would like you to address all issues raised by the reviewers in a revision.
Moreover, there were some confusions about pilot data and it was not clearly stated in the manuscript that data collection is ongoing. I found in the meta-data of your submission that your study is considered bias-control level 2. As this means that changes in the experimental design are not possible anymore, you need to discuss the points raised by the reviewers regarding design, add them as possible limitations, and be transparent about the current state of the study. By transparency I mean that you might add a section on "Proposed timeline" or however you want to name it, that is typically added to Registered Reports Stage 1, and add details about what was done when and what will be done when.
Thank you for considering PCI-RR! Looking forward to your revision.
Best regards, Christina Artemenko
Reviewed by anonymous reviewer 1, 21 Feb 2024
In this pre-registered study, Hsu and authors plan to investigate a very important and interesting question that pertains to the field of brain stimulation: dose-response of tDCS effects with motor learning and cortical excitability. While this this topic is of interest and the overall manuscript is well-written, I have some concerns about the complexity of the study design.
The incorporation of challenging physiological measures such as TEPS and MEPs from both hemispheres, alongside multiple motor learning assessments within a single experimental session, raises concerns about participant fatigue and its potential influence on motor performance and physiological measures. The time-consuming nature of tasks such as TMS-EEG setup and obtaining MEP measures for both hemispheres could lead to fatigue among experimenters and participants alike. Additionally, the extensive number of MEPs per time-point may be excessive for estimating cortical excitability.
Furthermore, while I believe your current design adequately addresses the question regarding the effect of tDCS dose on motor sequence learning, I am unclear about the integration of the concept of generalization into the project. It is essential to clarify the expected outcome and consider how the experimental procedures, including the measurement of MEPs and TEPs, following a break might influence aspects of motor consolidation and generalization.
Regarding the question of whether concurrent tDCS affects corticospinal excitability, I have reservations about the current design's ability to provide adequate testing. Combining tDCS with motor learning may saturate cortical excitability, making it challenging to isolate the effects of tDCS dosage on cortical excitability independent of motor learning.
To address these concerns, I suggest considering two separate experimental sessions: one focusing on recording physiological measures before and after tDCS administration (with groups separated by tDCS dose), and another session dedicated to testing the effects of motor learning. This approach would help mitigate issues related to fatigue and provide a clearer understanding of the independent effects of tDCS dosage on cortical excitability.
https://doi.org/10.24072/pci.rr.100656.rev11Reviewed by Charlotte Wiltshire , 05 Mar 2024
, 05 Mar 2024
This study aims to understand the neuromodulatory effects of HD tDCS at higher stimulation intensities on motor learning and cortical excitability. The design appears to be appropriate and well powered. Given the growing inconsistency in this field, this is a timely and interesting study! I do have a few comments and concerns. I would like to hear more about the safety protocols that are being used, particularly the interaction of tDCS and TMS protocols. I also have some concerns over the “pre-test” used to assign participants to the tasks. Please see comments below for details.
Rationale is good, though further discussion of the discrepancies between positive and negative findings would be useful. There is not much detail on what the scepticism around tDCS is based on, for example, and what this would mean for the field if there is no relationship between tDCS and cortical excitability. There are a couple of references to work that has not shown an effect of tDCS on motor cortex excitability but they are not discussed in detail and other work could be added to build a better picture of the current state of the field.
Experimental design, including researcher blinding is appropriate.
Will the time of day be set for all of the participants? Justification for/against? (Effects on GABA, wakefulness, caffeine, food).
Caffeine intake prior to the experiment should be monitored for both participant safety and the effect on the experiment. I suggest keeping caffeine intake in line with the participant’s normal dose, but no caffeine within 1-hr of testing.
The typing test:
I’m a little confused about this task. In the procedures section, it describes the task as typing real (English) words. In the “Predicting motor learning ability” section, it describes it as a series of letters. This distinction is important and relates to my next comments.
The typing test seems weighted towards those who have learned to touch-type, rather than capturing a general measure of de-novo sequence learning or baseline sequence speed. This may also be affected by the languages spoken by the participant. This would need consideration of the number of languages spoken and the proficiency of English, though I would prefer to see a test that is not 1) linked with language use and 2) linked with a skill that the participants may or may not have previously learned (touch typing). Similarly, performance on this task may rely on touch-typing abilities which are not easily transferable to number typing i.e., participants may have automatic, well learned behaviours for typing that are not apparent for pressing numbers on a response pad.
This also applies for the analysis plan. Including this as a co-variate in the model seems like you would capture variation in touch-typing proficiency rather than any baseline level of sequence execution.
After reading until the end, I see more justification for using this typing measure based on previous work. If this task is used, this justification needs to come earlier in the manuscript but my above concerns for this task still stand.
A clearer description of the benefits of this neuro-navigation procedure would be useful for those not familiar with this set-up. For example, this study does not use subject-level anatomy (MRI structural scan), and so is relying on hotspots to find the hand representation on the motor cortex. There’s no problem with this, but a little more explanation may be useful. The main benefit of neuro-navigation comes from being able to monitor the position of the coil (make sure it is consistent throughout the study in terms of angle and location) and being able to move the coil back to exactly the same place before and after training.
Why are 60 pulses used to assess the baseline MEP level? Standard protocols use ~20-30 MEPs. This saves time but is also important for participant safety. The number of pulses (from the very start of the hotspot/thresholding procedure) until the final pulse should be monitored and kept within safety operating procedures. Please state what these are for your institution/protocol. My reading is that you will have 60 MEPs for LH (Right FDI), 60 for RH (Left FDI), plus ~50 for “hotspotting” each side. How is this expected to interact with the particularly high doses of tDCS (up to 4 electrodes with 1.5mA)?
1cm steps for locating the hotspot seems large. Smaller increments may be necessary to find the best hotspot. Any variability in this between participants (e.g due to not being accurate on the hotspot) will lead to significant variability in MEP.
Please expand on the use of both left and right hemispheres. Presumably this is a control condition, but I don’t see this included in the analysis plan or explicitly mentioned in the design of the experiment. Apologies if I have missed this.
Blinding of the participants is not expected. Some more detail on the implications of this is important. Will participants have any prior experience with tDCS? If not, what evidence is there that the ramping procedure will not be an effective sham?
What is the rationale for using effect sizes in line with previous studies for H1 and then being more conservative for H2?
Will any data cleaning be performed on the MEP data. For example, if there is no clear sign of an MEP for a trial, will this be included or excluded? Will you check that participants are at rest (EMG signal) during the MEP recordings? Will trials with a excess tension be excluded?
More details of the current modelling would be useful. What was used to model current flow and density?
Only later on is it mentioned that a very similar study has been run (Pilot data). Is this a replication study or does this build on this work in some way? Perhaps putting this “pilot” study into the introduction would be appropriate. It seems as though more emphasis should be put on this prior study and the fact that this is a replication (or part-replication?).
What is the maximum for the sensation rating score used in the pilot experiment? This would help to interpret graph 3B. Are there any checks in place to make sure that participants tolerate the stimulation to an acceptable level? Participants will, of course, be told that they can stop at any time, but given the potential for adverse effects with higher doses, will there be any additional measures taken?
The analysis plan suggests that a few different null effects are predicted (e.g. a saturating effect; there will be no difference between the 4mA and 6mA conditions). Further investigation of this effect using Bayesian analysis may help to provide support in the event that there is a null finding. My impression of this work is that Bayesian Hierarchical regression modelling could be implemented in the same way as the linear models.
https://doi.org/10.24072/pci.rr.100656.rev12








